The federal regulations that inform the work of the IRB can be found in Title 45, Part 46 of the Code of Federal Regulations (45 CRF 46).įor information about what 45 CFR 46 is, see. Failing to maintain federal standards governing this protection-whether from researchers pursuing research without proper approval or from the IRB granting improper approval-can result in the loss of all federal research funding at the school. If the certification is not submitted within these time limits, the application or proposal may be returned to the institution.The federal government requires every institution that receives federal research funding to maintain an Institutional Review Board (IRB) for the protection of human subjects. Institutions without an approved assurance covering the research shall certify within 30 days after receipt of a request for such a certification from the Department or Agency, that the application or proposal has been approved by the IRB. Under no condition shall research covered by §46.103 of the policy be supported prior to receipt of the certification that the research has been reviewed and approved by the IRB. Such certification must be submitted with the application or proposal or by such later date as may be prescribed by the Department or Agency to which the application or proposal is submitted. An institution with an approved assurance shall certify that each application or proposal for research covered by the assurance and by §46.103 of this policy has been reviewed and approved by the IRB. “Certification is required when the research is supported by a Federal Department or Agency and not otherwise exempted or waived under §46.101 (b) or (i). Texas A&M International University IRB is also certified and is noted in the grant office: The exemption at 45 CFR 46.101(b)(2), for research involving survey or interview procedures or observation of public behavior, does not apply to research with children, Subpart D, except for research involving observations of public behavior when the investigator(s) do not participate in the activities being observed.” The exemptions at 45 CFR 46.101(b) do not apply to research involving prisoners, fetuses, pregnant women, or human in vitro fertilization, Subparts B and C. “Institutions with DHHS-approved assurances on file will abide by provisions of Title 45 CFR Part 46 Subparts A-D. Texas A&M International University IRB has the DHHS-approved assurances on file. The federal regulations require that IRBs give special consideration to protecting the welfare of vulnerable subjects, such as children, prisoners, pregnant women, mentally disabled persons, or economically or educationally disadvantaged persons. The OHSR operates within the National Institute of Health (NIH) and the guidelines are available on-line. 
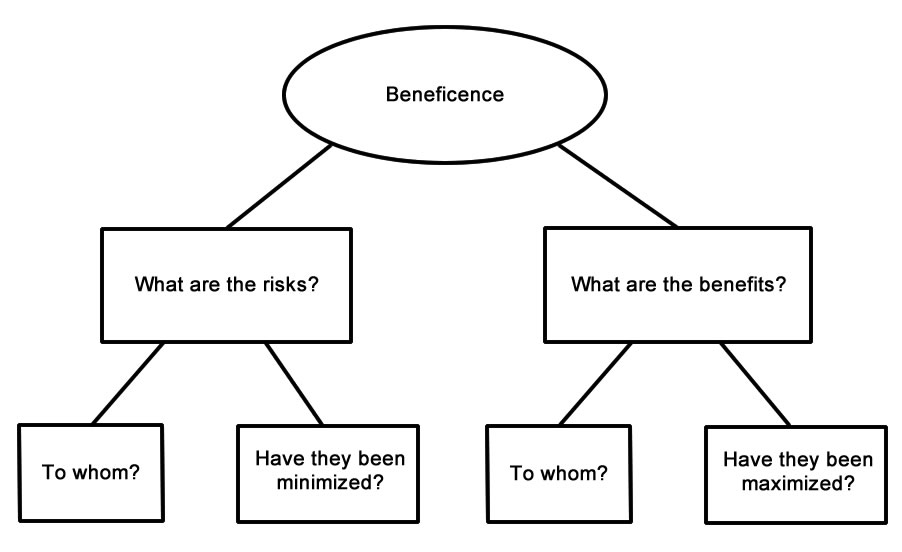
These regulations were revised in 1991 and are interpreted by the Office of Human Subjects Research (OHSR). The Department of Health and Human Services (DHHS, formerly DHEW) regulations are codified at Title 45 Part 46 of the Code of Federal Regulations. The principle of beneficence requires the researcher to do good and “above all, do no harm.” The principle of justice holds that human subject should be treated fairly. The principle of respect for persons holds that persons have the right to self-determination and the freedom to participate or not participate in research. The Commission developed The Belmont Report which identified three ethical principles as relevant to the conduct of research involving human subjects. In addition, the National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research (1978) was formed. The continued conduct of harmful, unethical research made additional controls necessary, in 1973, the Department of Health, Education, and Welfare Regulations (DHEW) published its first set of regulations on the protection of human subjects. The first provision of the Code states that “the voluntary consent of the human subject is absolutely essential.” See ( ). The Code captures many of what are now taken to be the basic principles governing the ethical conduct or research involving human subjects. 
Human subjects protection begins with the Nuremberg Code developed for the Nuremberg Military Tribunal as standards by which to judge the human experimentation conducted by the Nazis. HISTORY OF THE HUMAN SUBJECTS PROTECTIONS SYSTEM: Institutional Review Board (IRB) GuidelinesĪ. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
